Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
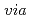 thermally induced phase separation
thermally induced phase separationSawada, Shinichi; Ursino, C.*; Galiano, F.*; Simone, S.*; Drioli, E.*; Figoli, A.*
Journal of Membrane Science, 493, p.232 - 242, 2015/11
Times Cited Count:53 Percentile:88.7(Engineering, Chemical)The replacement of commonly-used substances with non-toxic equivalents is attracting a great amount of attention in membrane preparation processes. In order to address this issue, we prepared porous poly(vinylidene fluoride) (PVDF) membranes via thermally-induced phase separation using the following non-toxic Citroflex as solvents: acetyl tributyl citrate (ATBC); acetyl triethyl citrate (ATEC); and triethyl citrate (TEC). The pore size of the membranes increased in the following solvent order of ATBC  ATEC
ATEC  TEC, which is the same trend of the PVDF/solvent affinity. During the phase separation process, high-affinity solvent molecules should have enough molecular mobility and easily gather with each other to form the large solvent rich phases, thereby producing the large pores. In the pure water microfiltration test using the PVDF membranes, the water permeability can be controlled in the wide range, depending on the pore size.
TEC, which is the same trend of the PVDF/solvent affinity. During the phase separation process, high-affinity solvent molecules should have enough molecular mobility and easily gather with each other to form the large solvent rich phases, thereby producing the large pores. In the pure water microfiltration test using the PVDF membranes, the water permeability can be controlled in the wide range, depending on the pore size.
Katayama, Yoshinori
Nihon Butsuri Gakkai-Shi, 60(6), p.456 - 460, 2005/06
By means of in-situ high-temperature and high-pressure experiment, we found two distinct structures of molten phosphorus, one is a molecular fluid and the other is a polymeric liquid, and an abrupt and reversible structural transformation between them at about 1 GPa. Further investigations revealed that the two structures have different densities and they separate macroscopically during the transformation. These results support a view that this change is an extremely rare example of a first-order phase transition between two stable disordered phases besides liquid-gas transition.
Kubo, Shinji; Yoshida, Mitsunori; Sakurai, Makoto*; Tanaka, Kotaro*; Miyashita, Reiko*
Bunri Gijutsu, 35(3), p.148 - 152, 2005/05
no abstracts in English
Koizumi, Satoshi
Journal of Applied Crystallography, 36(Part3,2), p.381 - 388, 2003/06
no abstracts in English
Miura, Takayuki*; Kuwano, Hisashi*; Kikuchi, Kenji
Tetsu To Hagane, 87(2), p.31 - 36, 2001/02
no abstracts in English
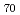 Ca
Ca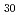 MnO
MnO
Katano, Susumu; Fernandez-Baca, J. A.*; Yamada, Yasusada
Physica B; Condensed Matter, 276-278, p.786 - 787, 2000/03
Times Cited Count:9 Percentile:47.89(Physics, Condensed Matter)no abstracts in English
Ebihara, Kenishi; Watanabe, Tadashi; Kaburaki, Hideo
Int. J.Modern Phys. C, 9(8), p.1417 - 1427, 1998/00
Times Cited Count:2 Percentile:20.33(Computer Science, Interdisciplinary Applications)no abstracts in English
Ebihara, Kenishi; Watanabe, Tadashi; Kaburaki, Hideo
JAERI-Research 97-043, 26 Pages, 1997/07
no abstracts in English
Ebihara, Kenishi; Watanabe, Tadashi; Kaburaki, Hideo
Dai-11-Kai Suchi Ryutai Rikigaku Shimpojiumu Koen Rombunshu, p.239 - 240, 1997/00
no abstracts in English
Ebihara, Kenichi; Watanabe, Tadashi;
Dai-10-Kai Suchi Ryutai Rikigaku Shimpojiumu Koen Rombunshu, p.280 - 281, 1996/12
no abstracts in English
; Kobayashi, Hiroaki; Masaki, Toshio; *; Kawamura, Kazuhiro; ;
PNC TN8440 94-028, 173 Pages, 1994/06
None
Nihon Kessho Gakkai-Shi, 26(1), p.87 - 99, 1984/00
no abstracts in English
;
Physical Review Letters, 52(10), p.835 - 838, 1984/00
Times Cited Count:137 Percentile:97.17(Physics, Multidisciplinary)no abstracts in English
; *; *
J.Chem.Eng.Jpn., 16(6), p.513 - 516, 1983/00
Times Cited Count:5 Percentile:69.88(Engineering, Chemical)no abstracts in English
;
Physica B; Condensed Matter, 120, p.392 - 396, 1983/00
no abstracts in English
;
Journal of the Physical Society of Japan, 51(2), p.347 - 348, 1982/00
Times Cited Count:13 Percentile:79.53(Physics, Multidisciplinary)no abstracts in English
 -amylase by Radiation-Induced Polymerization at Low-Temperature,III; Immobilization of High Concentration
-amylase by Radiation-Induced Polymerization at Low-Temperature,III; Immobilization of High Concentration  -amylase by the Polymerization of 2-Hydroxyethylmethacrylate
-amylase by the Polymerization of 2-Hydroxyethylmethacrylate; ; Kaetsu, Isao
JAERI-M 6190, 13 Pages, 1975/07
no abstracts in English
Sakamoto, Atsushi; Takeuchi, Masayuki; Ogino, Hideki; Koizumi, Kenji; Sawahata, Osamu*; Sakamoto, Yukio*; Akutsu, Koichi*
no journal, ,
no abstracts in English
 ,
, -dialkylamides-nitric acid systems by turbidity measurement, 2; The Correlation between turbidity and the distribution ratio of uranium
-dialkylamides-nitric acid systems by turbidity measurement, 2; The Correlation between turbidity and the distribution ratio of uraniumTsutsui, Nao; Ban, Yasutoshi; Hakamatsuka, Yasuyuki; Urabe, Shunichi; Matsumura, Tatsuro
no journal, ,
The turbidity of the agitated solutions of  ,
, -di(2-ethylhexyl)-butanamide (DEHBA) in dodecane and uranyl nitrate in 3 M HNO
-di(2-ethylhexyl)-butanamide (DEHBA) in dodecane and uranyl nitrate in 3 M HNO was measured for 30 min. The variation of the distribution ratio of uranium after stopping the agitation was also measured, and the correlation between the turbidity and the distribution ratio was studied. The turbidity and the distribution ratio of uranium at 3 min after stopping the agitation was more than 1000 FTU and 1.7, respectively, when the initial uranium concentration was 700 mM. The turbidity at 6 min decreased to 363 FTU, while the distribution rate increased to 2.2. Although the turbidity after 6 min decreased gradually, the distribution ratio barely changed. The correlation between the turbidity and the distribution ratio is discussed.
was measured for 30 min. The variation of the distribution ratio of uranium after stopping the agitation was also measured, and the correlation between the turbidity and the distribution ratio was studied. The turbidity and the distribution ratio of uranium at 3 min after stopping the agitation was more than 1000 FTU and 1.7, respectively, when the initial uranium concentration was 700 mM. The turbidity at 6 min decreased to 363 FTU, while the distribution rate increased to 2.2. Although the turbidity after 6 min decreased gradually, the distribution ratio barely changed. The correlation between the turbidity and the distribution ratio is discussed.
Sakamoto, Atsushi; Sano, Yuichi; Takeuchi, Masayuki; Ito, Kazuyuki*; Sekita, Satoshi*; Sakamoto, Yukio*; Akutsu, Koichi*
no journal, ,
no abstracts in English